
from the LSFC team and on the following topic:
"Acoustic cavitation contribution to the depollution by leaching of vermiculite contaminated with Cs, Ni or Zn".
Defense scheduled for Friday, June 2, 2023 at 9:30 am (ICSM Auditorium).
Owing to anthropogenic action and past and present industrial activity, a growing proportion of sites in France are polluted with radioelements and heavy metals, leading to a major risk for human health and the environment. In this context, the development of remediation techniques allowing the extraction of these pollutants is required for the rehabilitation of soils. Among existing methods, leaching is an effective process, simple to implement and with treatment kinetics compatible with industrial implementation. However, the results reported in the literature are still open to improvement. This thesis investigates the contribution of a process combining leaching with an acid and an ion exchanger and the use of ultrasound for the remediation of cesium and heavy metals on vermiculite clay, compared to conventional leaching processes.
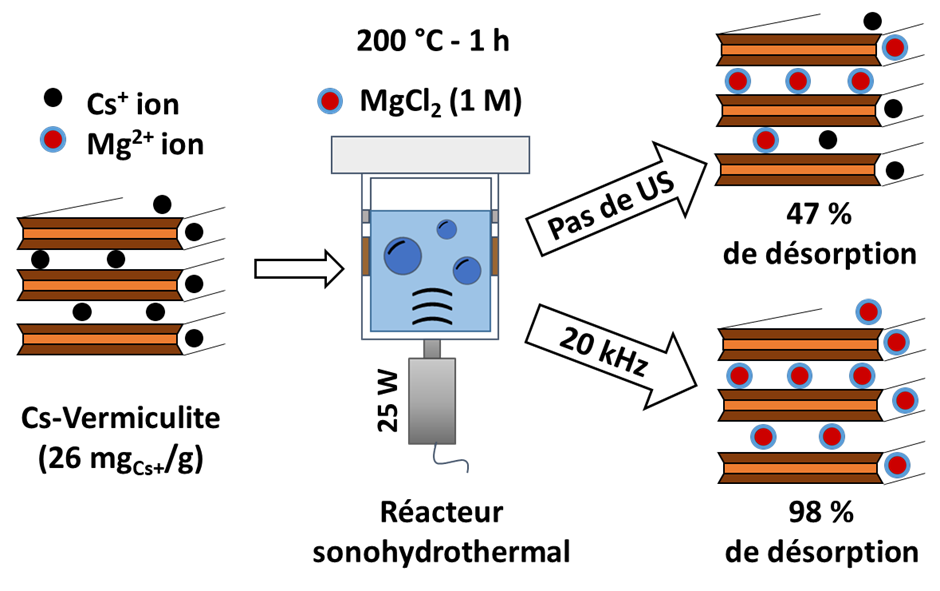
The treatment by ion exchange of a vermiculite contaminated with cesium was accelerated at room temperature in the presence of ultrasound but remained very limited. By coupling hydrothermal conditions (up to 200°C) to low-frequency ultrasound (20 kHz), a nearly complete desorption of Cs from the vermiculite could be achieved. The mechanism involved in the process was explained as an improvement in the accessibility of the cesium by expanding the interfoliar space, in which the contaminant is strongly fixed.
In the case of heavy metal polluted vermiculite, different effects were noticeable depending on the pollutant considered. The distribution of the pollutant in the different vermiculite fractions (exchangeable, carbonate bound, Fe/Mn oxide bound, organic bound and residual) was determined by sequential extraction. Zinc was mainly fixed in acid leachable fractions and could be quantitatively and rapidly desorbed by the use of an inorganic (HCl) or bio-sourced organic acid (citric acid). The contribution of ultrasound, regardless of the chosen frequency, was found to be insignificant compared to the influence of the acidity of the solution or the vermiculite mass/solution volume ratio. The desorption kinetics of Ni were slower than for Zn, resulting in limited desorption yields after three hours of treatment in either HCl or citric acid. The sequential extraction protocol attributed this tendency to the presence of Ni mostly on difficult to access sites. The use of ultrasound at a frequency of 362 kHz improved kinetics and desorption yields during the treatment in the presence of HCl.
Credit: S. Herr / ICSM
A major difference in behavior between low (20 kHz) and high (362 kHz) frequencies is the significant fragmentation of the particles in the former case, leading to the formation of new adsorption sites on which metallic elements can re-adsorb. To take advantage of this phenomenon, a cation exchange in the presence of an earth-alkaline cation Mg2+ can then improve the desorption yields by preventing the re-adsorption of Ni or Zn.
Keywords: Depollution, desorption, heavy metals, leaching, cavitation, vermiculite